Winning the SME Grand Slam of EU Funding
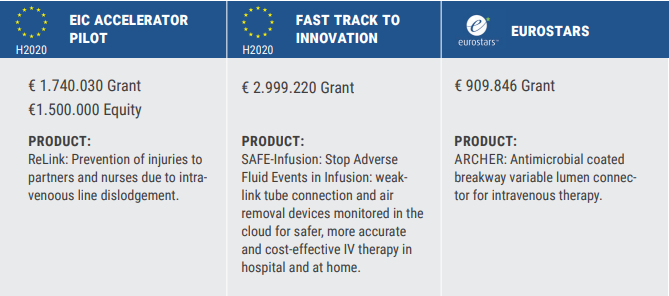
The Swedish medical device company, Tada Medical has successfully received grants from the three major EU funding programmes: EIC Accelerator, EIC Fast Track to Innovation (FTI) and Eurostars. It is a fantastic achievement as competition for grants has never been more intense. Moreover, Tada Medical is one of the handful SMEs that have accomplished this!
"The SME Funding Grand Slam" feels like a dream, but dreams do come true, says the CEO, Katarina Hedbeck. Tada Medical has succeeded in winning the Funding Grand Slam in just one year. They made it possible with their innovation ReLink, anchored on the disruptive approach to make IV therapy safer by preventing IV cannula dislodgement.
Successful funding process
A great innovative business idea is obvious for starting the funding proposal process. However, having the right idea does not ensure success as the competition for funding is so increasingly strong! To be successful in the highly competitive and attractive grant funding process, you need a well written and structured project proposal that stands out from the crowd.
“We quickly realised that preparing and writing a compelling grant proposal is a challenging and a very specialised task. However, our own robust determination and belief in our team together with the right competencies, knowledge and expertise from the grant consultancy, Nordic Innovators, we achieved the most difficult targets with seamless ease,” says Katarina Hedbeck.
“Competition for EU grants has never been more intense! The funding success rate was 1.37% for the EIC Accelerator March 2020 cut-off, 1.95% for the FTI October 2020 cut-off and 19.88% for the Eurostars-2 February 2021 cut-off so we are very proud of the big achievement for Tada Medical”.
Improving lives with innovative solutions
Tada Medical has persevered to stay focussed on their vision to improve lives by providing innovative solutions for unmet healthcare needs. Their technology, the breakaway connector ReLink, includes - a weak link concept to prevent cannula dislodgement, self-sealable valves to prevent spill of drugs and blood, and reconnectable parts for rapid reinstatement of therapy. ReLink is the enabling platform technology supporting a family of five products, each targeting a specific niche. Collectively, they provide for a wide portfolio of large scale, global and profitable markets for human and animal care, in hospital and homecare settings.
Tada Medical’s next priorities are commercialisation of ReLink Care, product development for specific markets such as oncology and homecare, establishing low volume manufacturing setup and initiation of product development pipeline – targeting large sub-markets.











